Quality and Safety Assurance Framework
In order to provide customers with safe and reliable products, we have set our own strict standards for guaranteeing quality and safety in every process.
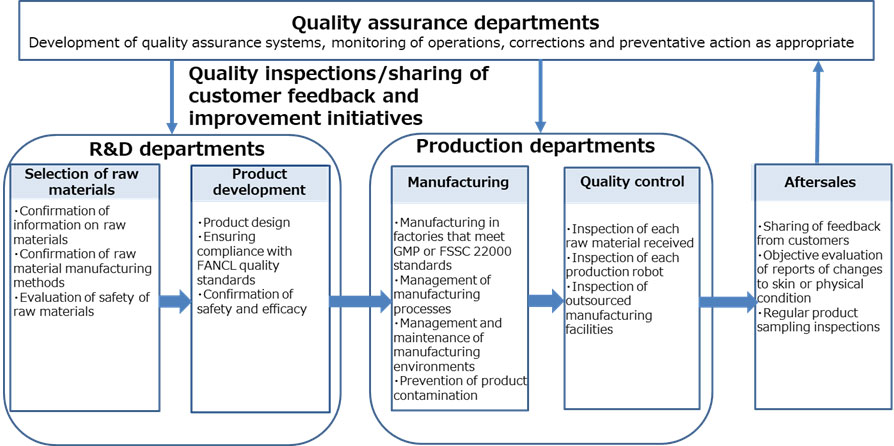
1. Selection of Safe Raw Materials

For all new raw materials that are considered for use in our products, we first verify their safety and evaluate their quality, and the desicion on whether to use them is made at a meeting attended by relevant departments.
- Verification of Raw Material Information
We investigate relevant regulations, raw material information, manufacturing methods, literature, and hazardous substances to confirm their safety. - Quality Evaluation
Depending on the characteristics of the raw materials, we conduct necessary tests, such as microbiological testing, testing for hazardous metals, allergens, and pesticide residues.
2. Product Development Focuses on Quality and Safety

We ensure that our products meet FANCL's unique quality standards, designed to provide our customers with peace of mind and safety.
- Cosmetics: Bringing natural skin beauty to our customers
- Free from preservatives, synthetic fragrances, synthetic dyes, petroleum-based surfactants, and UV absorbers
- Product design that minimizes skin irritation
- Safety verification tailored to product design (cytotoxicity testing, sensitive skin patch testing, sensitive skin usage testing, allergy testing, non-comedogenic testing, mild irritation testing, and usage testing supervised by dermatologists)
- Health Foods: Making products that prioritize ease of consumption and quality
- Formulation design focused on ease of consumption
Unique design prioritizes ease of consumption, including the thickness, roundness, size, taste, and smell of supplements. - Appropriate shelf-life setting
Shelf-life is set appropriately based on quality stability testing. - Safety confirmation through clinical trials
In cases where the safety data is insufficient, we conduct clinical trials to confirm that product consumption does not cause any changes in physical condition.
- Formulation design focused on ease of consumption
3. Maintaining a Sanitary Manufacturing Environment

At our six in-house production facilities across Japan for cosmetics and health foods, we manufacture products in accordance with each of the relevant standards as well as our own voluntary specifications, adhering to FANCL's strict proprietary standards with a mission to deliver safe and reliable products to our customers.
- Production at factories compliant with GMP and FSSC certification※
- Management of manufacturing processes
- Establishment and maintenance of a sanitary manufacturing environment
Establishing systems to prevent the introduction, generation, accumulation, and presence of dust and foreign matter. - Prevention of foreign matter contamination in products
The wearing of dedicated work uniforms thoroughly eliminates foreign matter such as dust and debris.
Access control through biometric authentification prevents intentional contamination.
Factory management focuses on food defense in health food manufacturing - Education and Training
We conduct regular employee training and quality control (QC) activities.
Cosmetics GMP compliant: Chiba Plant, Shiga Plant, Gunma Plant
Health Food GMP certified: Yokohama Plant, Mishima Plant
FSSC 22000 certified: Nagano Plant, Mishima Plant
Improving the safety of health foods: Manufacturing controls that incorporate food defense
We are working to prevent the intentional contamination of food products by establishing and constantly enhancing food defense systems. This includes introducing biometrics to limit access to manufacturing areas, installing cameras in manufacturing areas, requiring specified clothes to be worn, and controlling items that are allowed to be brought in.
- Only specified personnel are allowed to enter our factories. Furthermore, some of our factories use biometrics to control and record who enters production zones.
- We have installed cameras within factories so we can confirm the movement of people and products.
- We have made rules that limit the items that can be brought into production sites.
- Poisonous substances are kept locked up and thoroughly managed, and this management is carried out by a limited number of people.
- We provide employee education to raise awareness regarding food defense.
4. Thorough quality control to ensure safety and reliability

We have established robust inspection systems for incoming raw materials, in-process manufacturing, and our quality control department. Based on these systems, we determine whether products can be released for shipment.
- Raw material acceptance inspections
Verification of manufacturers' certificates of analysis
Visual and physical inspections, including checks for foreign matter contamination
Microbiological testing and component analysis
Expiration date management to prevent the use of degraded materials - Product inspections
In-process inspections to detect abnormalities early and prevent defective products from reaching the market
Quality control inspections to block shipment of products that do not meet strict standards for appearance, properties, microbiological quality, and absence of foreign matter - Audits of contracted manufacturing facilities
Regular site visits conducted
Verification of quality system effectiveness, manufacturing and QC practices, and employee training programs in line with GMP※ and other standards.
Good Manufacturing Practice: Standards for production management and quality control.
5. To ensure safe and reliable use for our customers

Fulfilling our mission to support customers' beauty and health, we continue to respond carefully and attentively to any concerns regarding our products.
- Sharing customer feedback internally
- Assessment of quality-related inquiries
Specialized departments promptly evaluate and review any potential link to the product, using findings as important insights for quality improvement - Assessment of reported skin reactions
Designated cosmetic safety experts ("Safety Management Officers") evaluate and review any potential link to the product - Assessment of reported health condition changes
A team of specialists, including medical professionals, evaluates and reviews any potential link to the product - Regular random product testing (spot checks)
